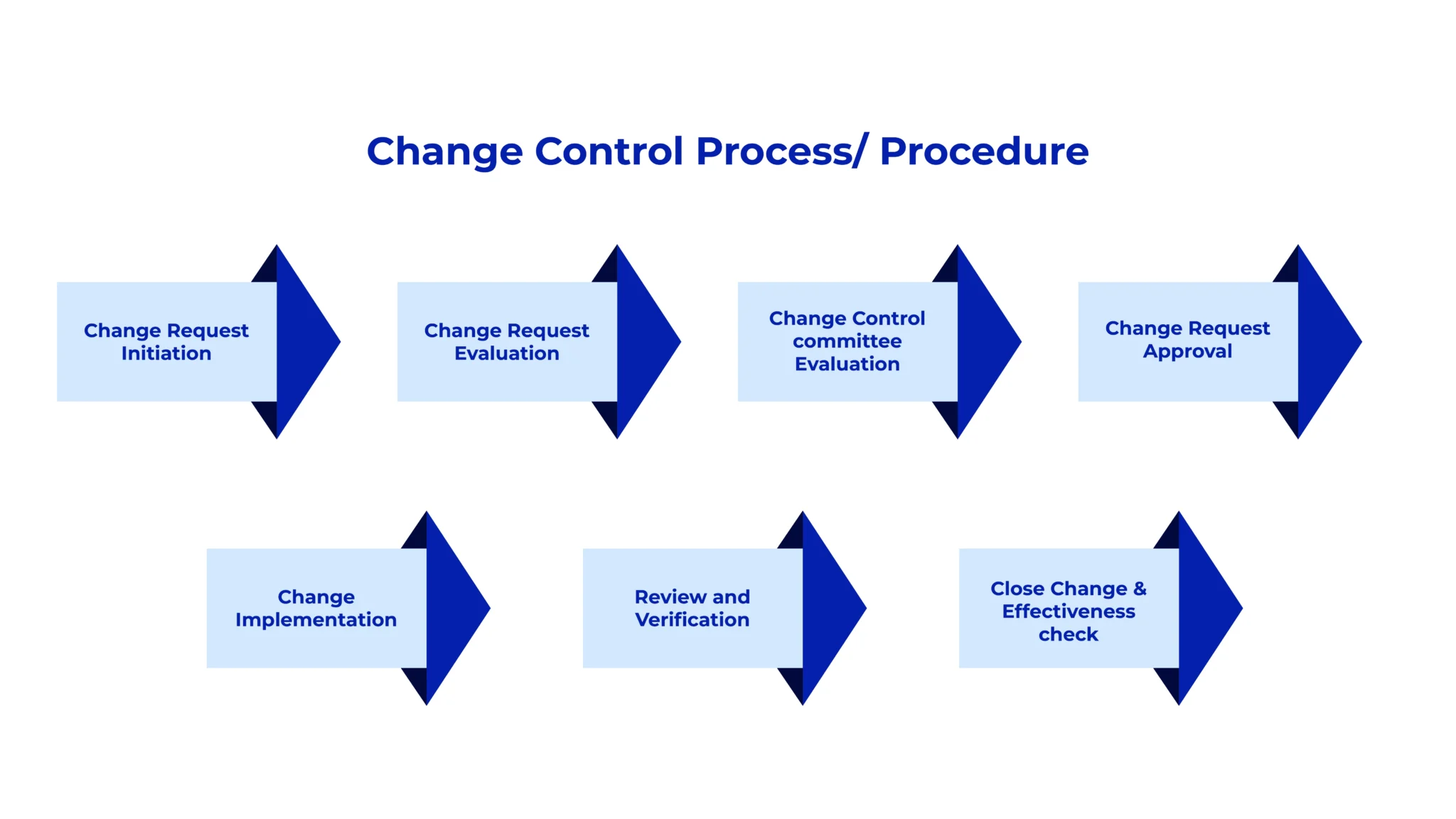
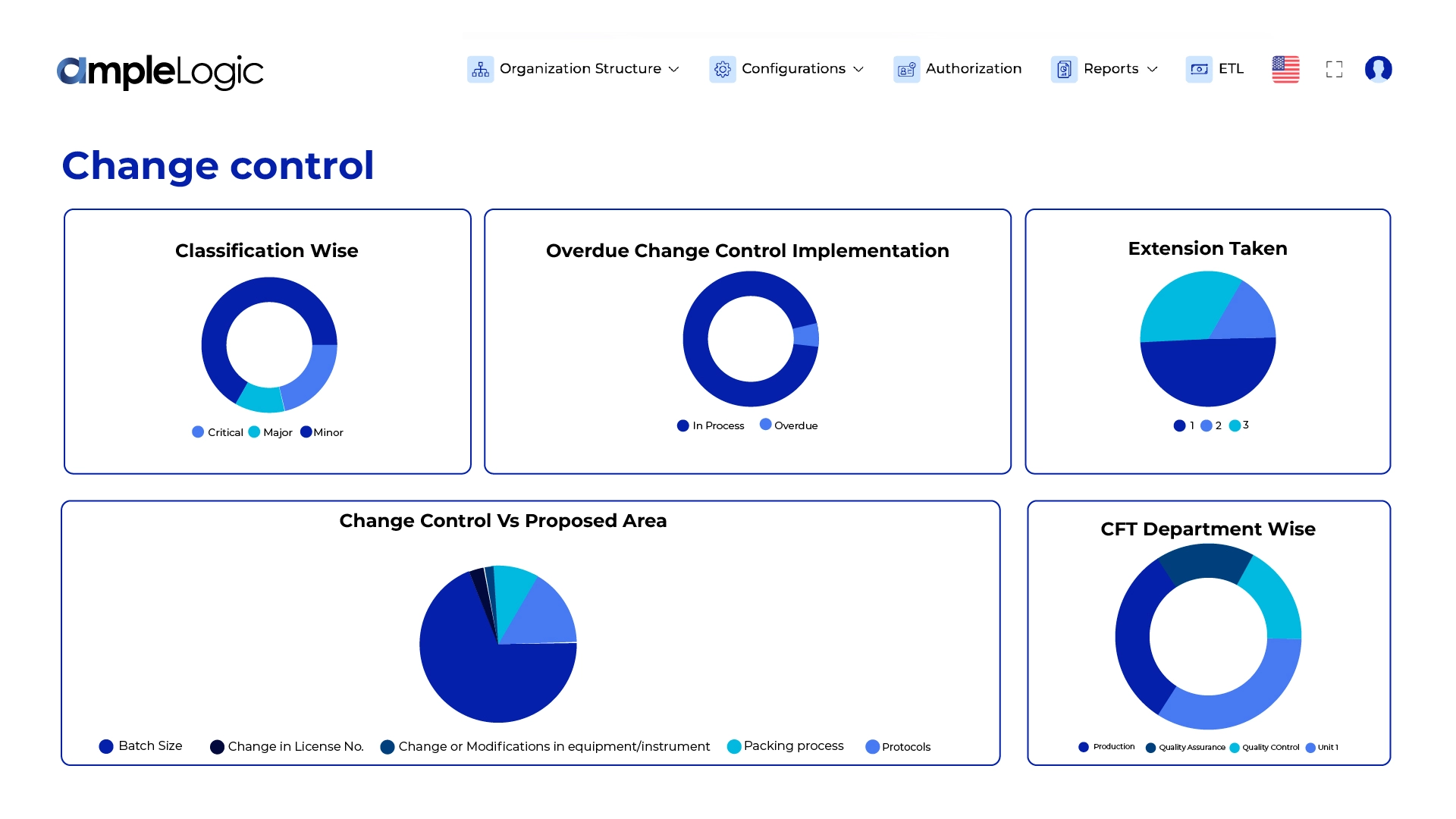
GAMP-Compliant AI Platform for Life Sciences & Regulated Industries
- GAMP Solutions
-
-
Pharma GAMP Solutions
APQR Annual Product Quality Review CAPS Calibration & Preventive Maintenance eQMS Electronic Quality Management System CVS Cleaning Validation SoftwareeLogBook Electronic Logbook System RIMS Regulatory Information Management System UAM User Access Management System PVS Process Validation Software
-
-
- Low-code aPaaS
- Resources
-
- Company
-
- Careers
Region
-
- GAMP Solutions
-
-
Pharma GAMP Solutions
APQR Annual Product Quality Review CAPS Calibration & Preventive Maintenance eQMS Electronic Quality Management System CVS Cleaning Validation SoftwareeLogBook Electronic Logbook System RIMS Regulatory Information Management System UAM User Access Management System PVS Process Validation Software
-
-
- Low-code aPaaS
- Resources
-
- Company
-
- Careers
Region
-